Finding Kinetic Arrhenius Parameters Using Parameter Estimation
Application ID: 10305
This example shows how to use the Parameter Estimation and Experiment features in the Reaction Engineering interface for optimization with multiple experimental data input files.
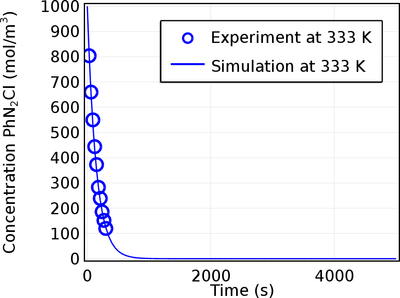
The application finds the Arrhenius parameters of a first order reaction where Benzene diazonium chloride decomposes to benzene chloride and nitrogen at 313, 319, 323, 328, and 333 K.
This model example illustrates applications of this type that would nominally be built using the following products:
however, additional products may be required to completely define and model it. Furthermore, this example may also be defined and modeled using components from the following product combinations:
The combination of COMSOL® products required to model your application depends on several factors and may include boundary conditions, material properties, physics interfaces, and part libraries. Particular functionality may be common to several products. To determine the right combination of products for your modeling needs, review the Specification Chart and make use of a free evaluation license. The COMSOL Sales and Support teams are available for answering any questions you may have regarding this.
